And so it is on to the Nobel Prize announcement for Chemistry and I’m betting that they have won the prize for doing something that is basically described using a collection of words you have never heard of and have no idea what they mean at all … well it is not just you, many often scratch their heads at such terms, but if you take the time to dig into it and decrypt, then you can indeed often find something fascinating.
And so it is on to the Nobel Prize announcement for Chemistry and I’m betting that they have won the prize for doing something that is basically described using a collection of words you have never heard of and have no idea what they mean at all … well it is not just you, many often scratch their heads at such terms, but if you take the time to dig into it and decrypt, then you can indeed often find something fascinating.
And so the 2014 prize has indeed been awarded for … (drum roll here please) … the development of super-resolved fluorescence microscopy
Yep, see what I mean – it’s word soup. OK, so what is this actually all about?
Seeing stuff that is very very small was once thought to have a hard limit, specifically half the wavelength of light, and so it appeared to be obvious that you could not possibly see something that needed a greater degree of resolution. Well these smart guys figured out clever ways to break the microscope barrier and have opened up new windows using what is perhaps best described as nanoscopy, via processes that allows us to see the pathways of individual molecules inside living cells. What can now be actually seen is truly amazing stuff, and can for example be utilised to see how molecules create synapses between nerve cells in the brain, or track proteins involved in Parkinson’s, Alzheimer’s and Huntington’s diseases as they aggregate, or follow individual proteins in fertilized eggs as these divide into embryos.
OK, so how does it all work?
Well, it turns out that two distinctly different ways of doing this are in the mix here.
Method 1
This one, known as stimulated emission depletion (STED) microscopy, was developed by Stefan Hell in 2000. Two laser beams are utilized; one stimulates fluorescent molecules to glow, another cancels out all fluorescence except for that in a nanometre-sized volume. Scanning over the sample, nanometre for nanometre, it yields an image
Method 2
Eric Betzig and William Moerner, working separately, laid the foundation for the second method, single-molecule microscopy. The method relies upon the possibility to turn the fluorescence of individual molecules on and off. Scientists image the same area multiple times, letting just a few interspersed molecules glow each time. Superimposing these images yields a dense super-image resolved at the nanolevel. In 2006 Eric Betzig utilized this method for the first time.
And here are the heroes themselves …
The Nobel Prize in Chemistry 2014 was awarded jointly to Eric Betzig, Stefan W. Hell and William E. Moerner “for the development of super-resolved fluorescence microscopy”.
| Eric Betzig | Stefan W. Hell | William E. Moerner |
Further Information
- Popular Information
- Scientific Background
- Image – Abbe’s diffraction limit (pdf 447 kB)
- Image – The principle of STED microscopy (pdf 369 kB)
- Image – The principle of single-molecule microscopy (pdf 2.2 MB)
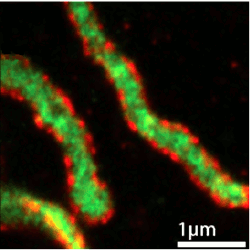
An example
Here is an image that shows you microtubules inside a living PtK2 cell …
Or how about actually seeing the distribution of membrane (red) and matrix (green) proteins in cellular mitochondria …